Your global partner for
end-to-end serialization
The global pharmaceutical industry faces major regulatory changes to improve supply chain security and patient safety. SCW partners with your organization to provide complete, cost-effective Track and Trace support across every major market and mandate.
Complete end-to-end serialization support
With project experience across all major serialization mandates, SCW guides pharmaceutical manufacturers from initial regulatory assessment through full operational compliance, whether you are approaching a first-time implementation or managing a complex multi-market program.
Our consultants bring hands-on expertise across L1 to L5 systems, CMO and trade partner onboarding, validation, daily operational support, and performance management, covering every layer of the serialization landscape.
Talk to an expert
Track & Trace service offerings
Our Track and Trace practice covers the full serialization lifecycle across 8 specialist areas, from regulatory intelligence and vendor selection through turnkey implementation, GAMP-5 validation, daily operational support, performance dashboards, training, and medical device compliance. Select a category below to explore the services within it.

Regulations
Staying ahead of global serialization mandates is one of the most critical and time-consuming challenges for pharmaceutical companies. Our regulatory intelligence service ensures your organization always has an accurate, up-to-date view of requirements across every market you operate in, turning compliance from a reactive burden into a proactive strategic capability.


Vendor Selection
Selecting the right serialization technology partner is a high-stakes decision with long-term implications for compliance, cost, and operational flexibility. SCW brings structured evaluation frameworks and deep market knowledge to ensure you choose solutions that fit your business today and scale as regulations evolve, without overspending or locking you into inflexible platforms.



Implementation
Implementation is where serialization programs succeed or fail. SCW's implementation services span the full delivery lifecycle, from line-level integration to global partner onboarding and cross-site standardization. We coordinate every workstream from vendor integration to compliance documentation, so your serialization infrastructure is compliant, scalable, and operationally resilient from day one.







Validation
Validation is a non-negotiable requirement for serialization systems in regulated pharmaceutical environments. SCW delivers rigorous, audit-ready validation documentation across every layer of your serialization architecture, from code print accuracy at L1 through to EPCIS message integrity at L5, giving regulators confidence and your operations the certainty to move at speed.


Daily Serialization Support
Serialization does not stop at go-live. The real operational complexity begins once systems are live and the daily volume of alerts, data queries, and compliance events requires ongoing expert attention. SCW provides on-call serialization support that keeps your operations running smoothly and your compliance posture intact every single day, so your team can focus on throughput rather than firefighting.





Performance Management
Most serialization programs meet functional requirements but leave reporting and analytics as an afterthought. SCW bridges this gap by connecting your L1-5 systems into dynamic dashboards that give operational teams real visibility into product movement, data quality, and line-level performance, enabling root cause analysis and continuous improvement across sites and CMO networks.


Training
Technology is only as effective as the people using it. SCW's serialization training programs develop competence and confidence across plant, warehouse, and quality teams, reducing human-error rejects, minimizing audit findings, and ensuring your teams can independently own compliance across the serialization landscape without relying on external support indefinitely.



Medical Device
Medical device manufacturers face their own distinct set of tracking and traceability requirements across the US and EU, governed by UDI mandates, EUDAMED registration obligations, and market-specific hub submission requirements. SCW's Medical Device practice applies the same depth of serialization expertise to the device sector, helping manufacturers navigate these mandates confidently and without disrupting commercial operations.




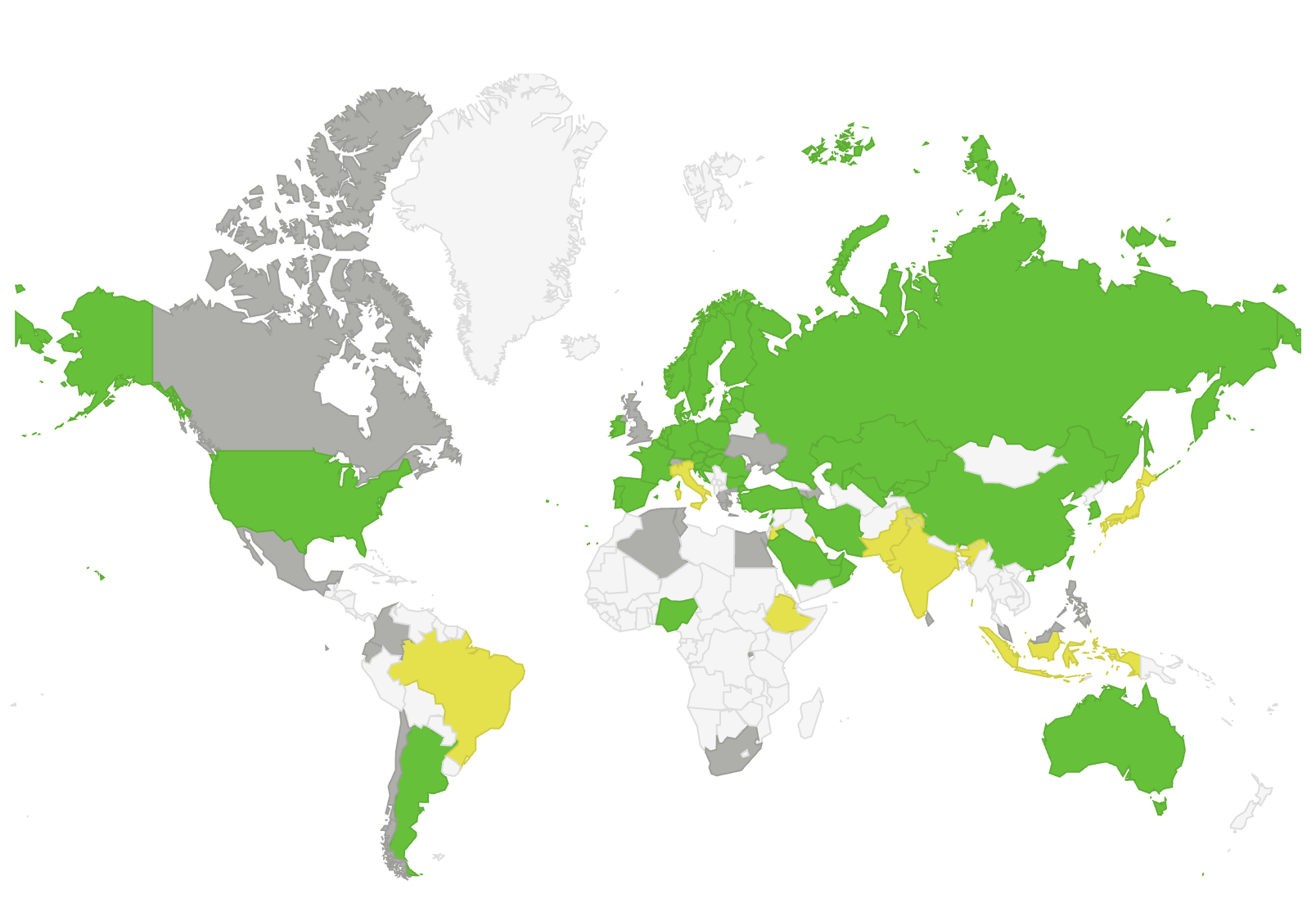
Global serialization regulation intelligence in one place
This database is prepared and maintained by Supply Chain Wizard to summarize Serialization, Track & Trace, and related regulations around the world for strategic planning purposes.
Ever-changing requirements, timelines, and milestones for serialization legislation are tracked globally and reported by country or region in an easy-to-understand and organized structure.
With comments, feedback, and questions from clients and users, the database and related services continue to grow stronger every day. We encourage users to interact with us through the comment and feedback functionality under each respective regulation for continuous improvement.
SCW supports companies on the long serialization program journey toward cost-effective compliance with end-to-end consulting services and specialized software solutions.
Prepare for the NDC12 transition before the compliance deadline
The National Drug Code is moving toward a standardized 12-digit format for drug products regulated by the U.S. FDA. The target compliance date is Mar 07, 2033, with no known exceptions currently.
The transition affects manufacturers, contract packaging organizations, wholesalers, distributors, pharmacies, healthcare providers, and health systems. It also creates downstream work across product labeling, packaging configuration, barcode data, system alignment, data governance, and training.
Track & Trace in practice
Real serialization results across global pharmaceutical manufacturers and contract organizations.
Ready to achieve global serialization compliance?
Tell us where you are in your Track and Trace journey and we will build a plan to get you compliant, fast.
Contact Us



